 Similarly, the p block are the right-most six columns of the periodic table, the d block is the middle 10 columns of the periodic table, while the f block is the 14-column section that is normally depicted as detached from the main body of the periodic table. Because of this, the first two rows of the periodic table are labeled the s block. If we use the periodic table as a guide, we can predict that two of the electrons would reside in the 4s orbital, and two of the electrons would reside in the 3d orbital. An atom with one or two valence electrons more than a closed shell is highly reactive, because the extra valence electrons are easily removed to form a positive ion. Titanium (Ti) is in group 4, thus it has 4 valence electrons. An atom with a closed shell of valence electrons (corresponding to an electron configuration (s2p6)) tends to be chemically inert. The periodic table, electron shells, and orbitals. The first two columns on the left side of the periodic table are where the s subshells are being occupied. Write the valence electron configuration of Ti and Ti 1+. Atomic structure and electron configuration. The ten-column block between these columns contains the transition metals. The first two columns on the left and the last six columns on the right as mentioned earlier are the main group elements. 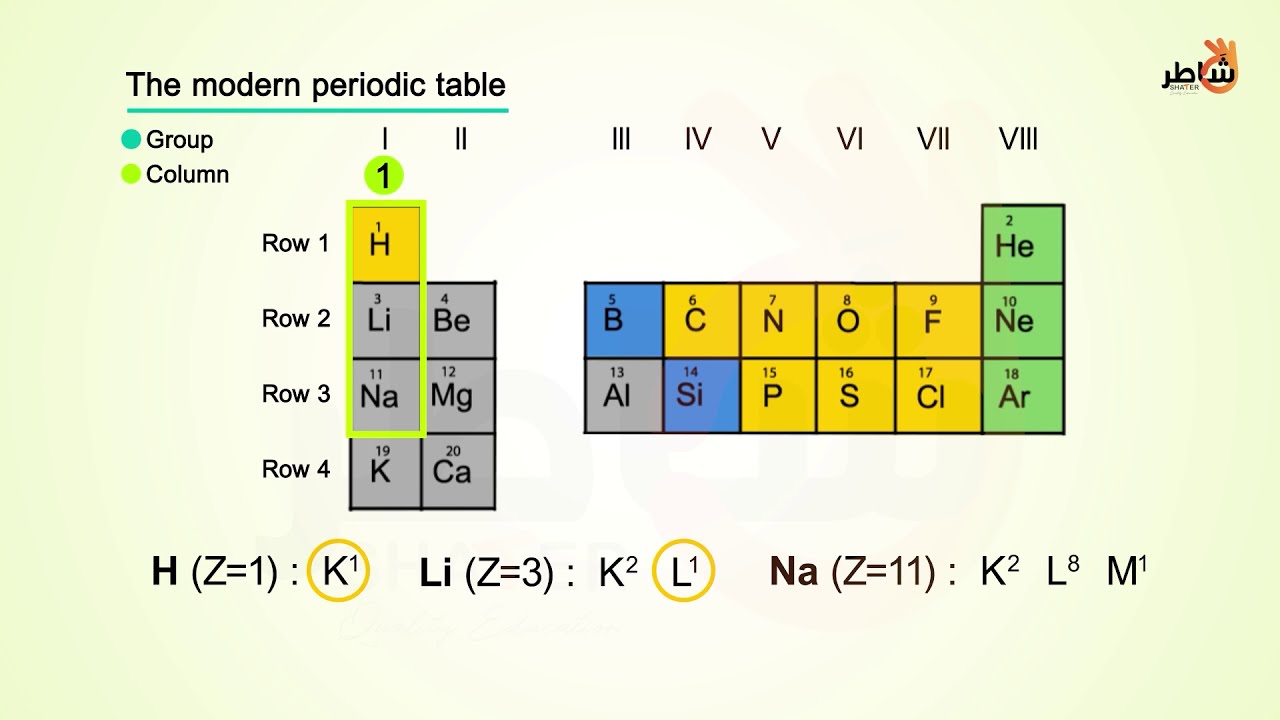
As we go across the columns of the periodic table, the overall shape of the table outlines how the electrons are occupying the shells and subshells. One way to categorize the elements of the periodic table is shown in Figure 3.8.1 3.8. The 3 d subshell is filled in the middle section of the periodic table.Īnd so forth. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
